全球首个、同类最优
新药研发



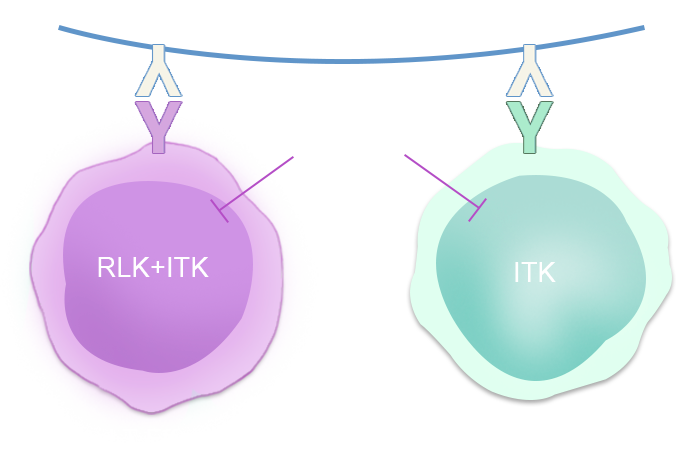
CPI-818是全球首款也是目前唯一进入临床阶段的ITK抑制剂,ITK(白细胞介素2诱导型T细胞激酶)是一种主要在T细胞上表达的酶,在T细胞和自然杀伤 (NK) 细胞淋巴瘤和白血病以及正常免疫功能中发挥作用。抑制T细胞中的特定分子靶标可能对于患有癌症(包括实体瘤)的患者以及患有自身免疫和过敏性疾病的患者具有治疗益处。对CPI-818作用机制的研究表明,它有潜力控制正常T辅助细胞的分化,并通过增强细胞毒性杀伤T细胞的产生和抑制癌细胞存活的细胞因子的产生来增强免疫反应。目前CPI-818已经获得FDA批准进入临床III期针对复发难治型外周T细胞淋巴瘤,其前期系列研究成果分别发表于美国癌症研究协会(AACR)年会(2023年4月)、第17届国际恶性淋巴瘤会议(ICML)(2023年6月)和第65届美国血液学会(ASH)年会(2023年12月)。
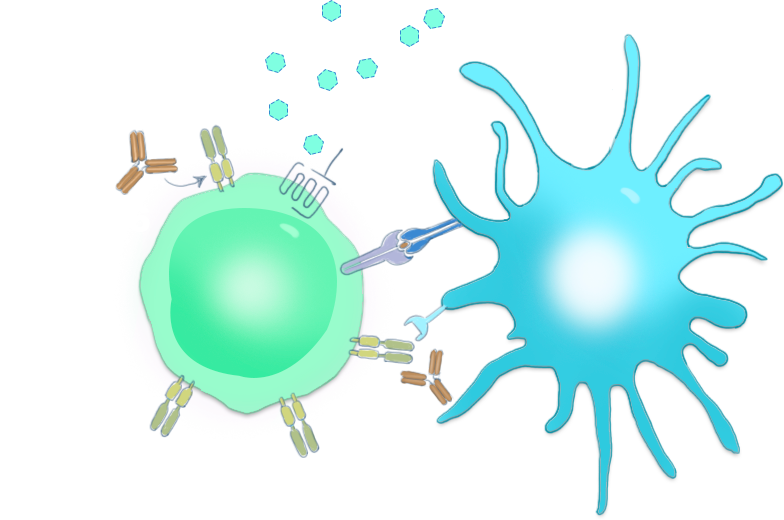
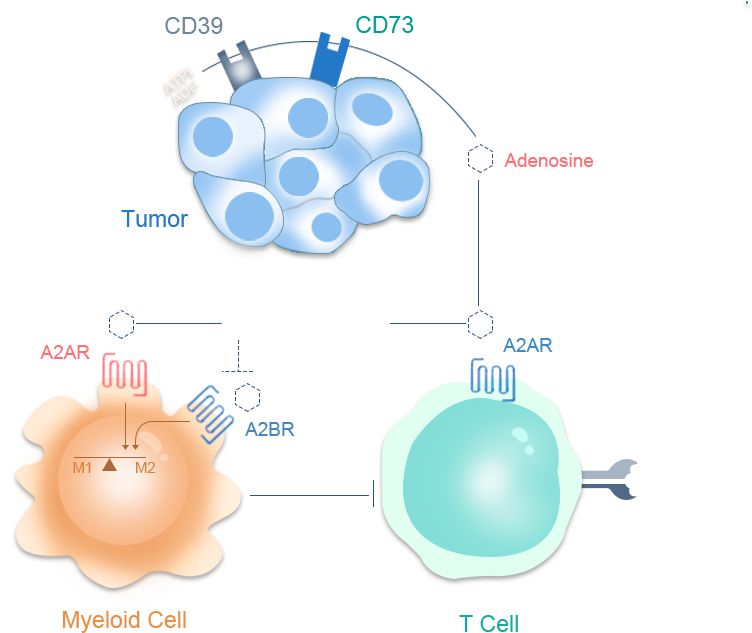
CPI-006是一种针对CD73的人源化单克隆抗体,具有激活B细胞以产生对肿瘤抗原和病毒的免疫反应的独特机制。在临床前研究中,已证明其免疫调节活性可导致淋巴细胞活化,并诱导B细胞产生抗体和影响淋巴细胞运输。与其他正在开发中的抗CD73抗体和小分子药物相比,CPI-006可刺激B细胞并阻断免疫抑制性腺苷的产生。B细胞的激活有可以增强肿瘤微环境内的免疫反应,从而改善临床结果。目前,中国区CPI-006联合帕博丽珠单抗针对非小细胞肺癌以及头颈鳞癌的临床试验正在进行,其中,CPI-006单药治疗非小细胞肺癌获得了非常出色的疗效数据。
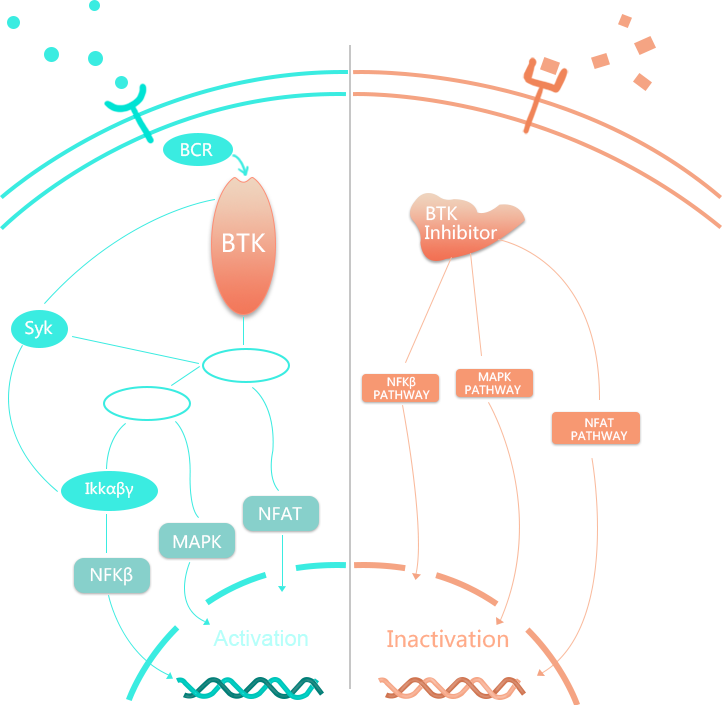
ANG-0623属于新一代高选择性的BTK抑制剂,研究揭示了其具有极佳的穿透血脑屏障特性,与其它BTK抑制剂相比,同样的剂量ANG-0623在动物模型脑脊液里的药物浓度远高于颈动脉中的药物浓度,,因此其对于原发性中枢神经系统淋巴瘤、多发性硬化症等中枢神经系统(CNS)疾病的治疗具有独特的潜在优势。目前ANG-0623已取得美国FDA和中国CDE针对多发性硬化症的临床试验I期批件。
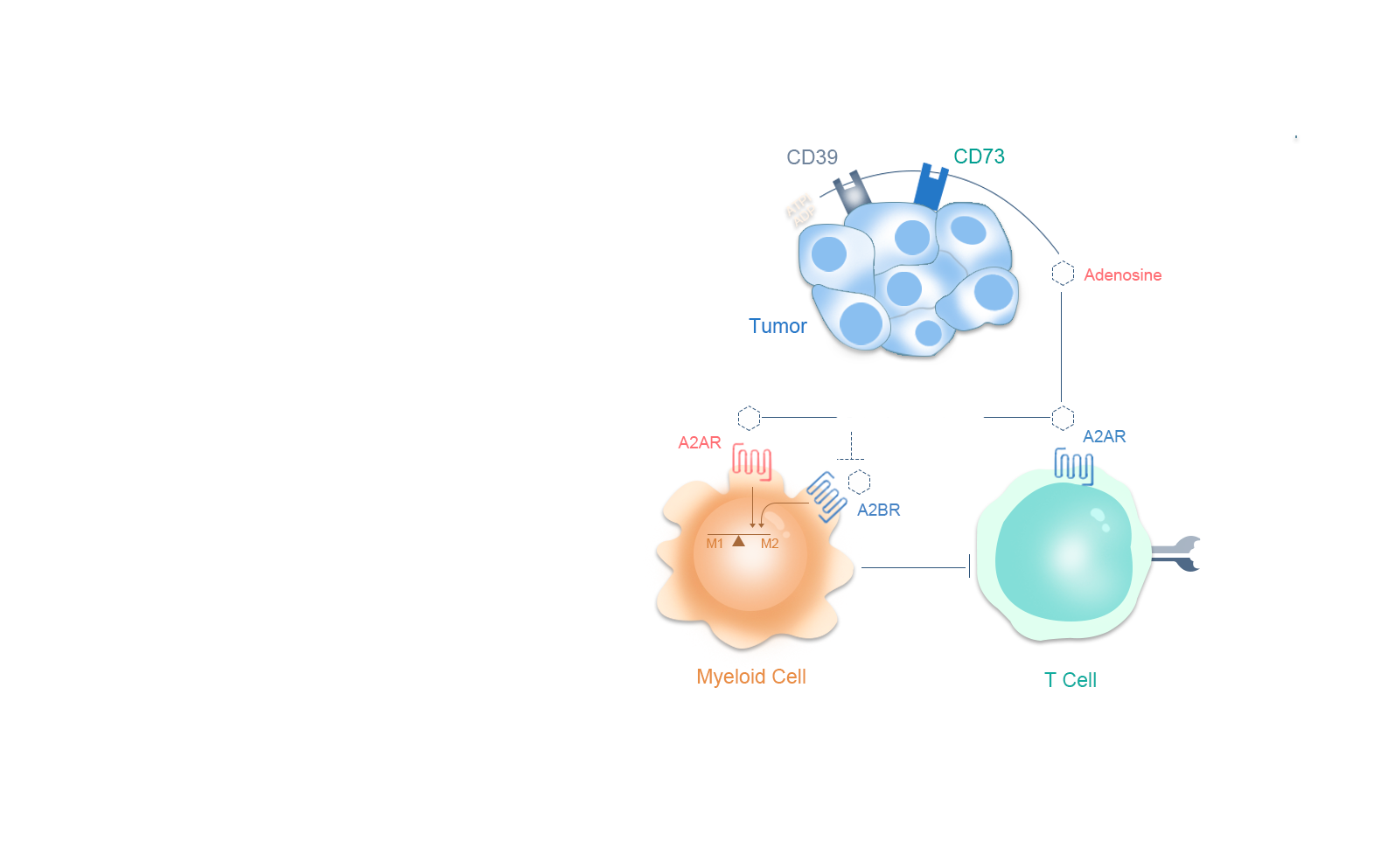
Ciforadenant是一种小分子腺苷A2A受体拮抗剂,是导致肿瘤微环境中免疫抑制的腺苷途径中的关键一步。I/Ib期临床试验的结果显示,Ciforadenant单独使用或与阿特珠单抗联合使用对晚期、难治性肾细胞癌患者有效。该研究还描述了腺苷基因标记,这是一种新的生物标志物,被发现有助于识别最有可能对治疗有反应的患者。和剂药业的合作伙伴Corvus正在与肾脏癌研究联盟(KCRC)合作,进行Ciforadenant联合伊匹单抗(抗CTLA-4)和纳武利尤单抗(抗PD-1)作为转移性肾细胞癌一线治疗的开放标签Ib/II期试验。试验设计基于Corvus在2018年发表在《癌症免疫学研究》(Cancer Immunology research)上的临床前研究,该研究表明使用Ciforadenant与抗CTLA-4和抗PD1联合使用在几个动物模型中显示出显著的抗肿瘤控制和治愈效果。